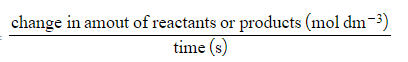Edexcel A Level Chemistry:复习笔记5.5.6 Reaction Order Graphs
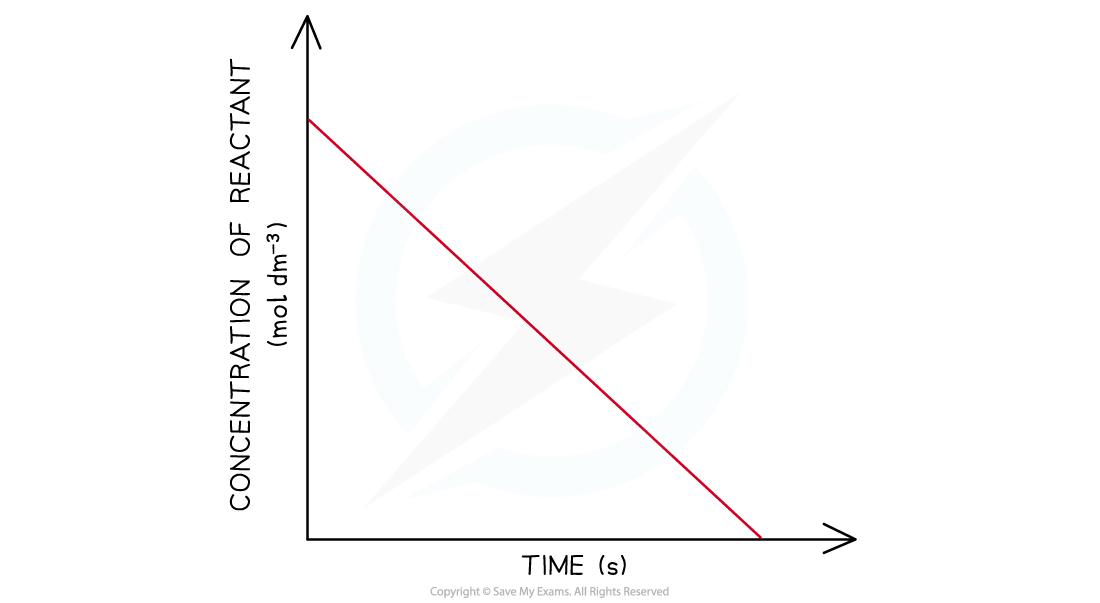
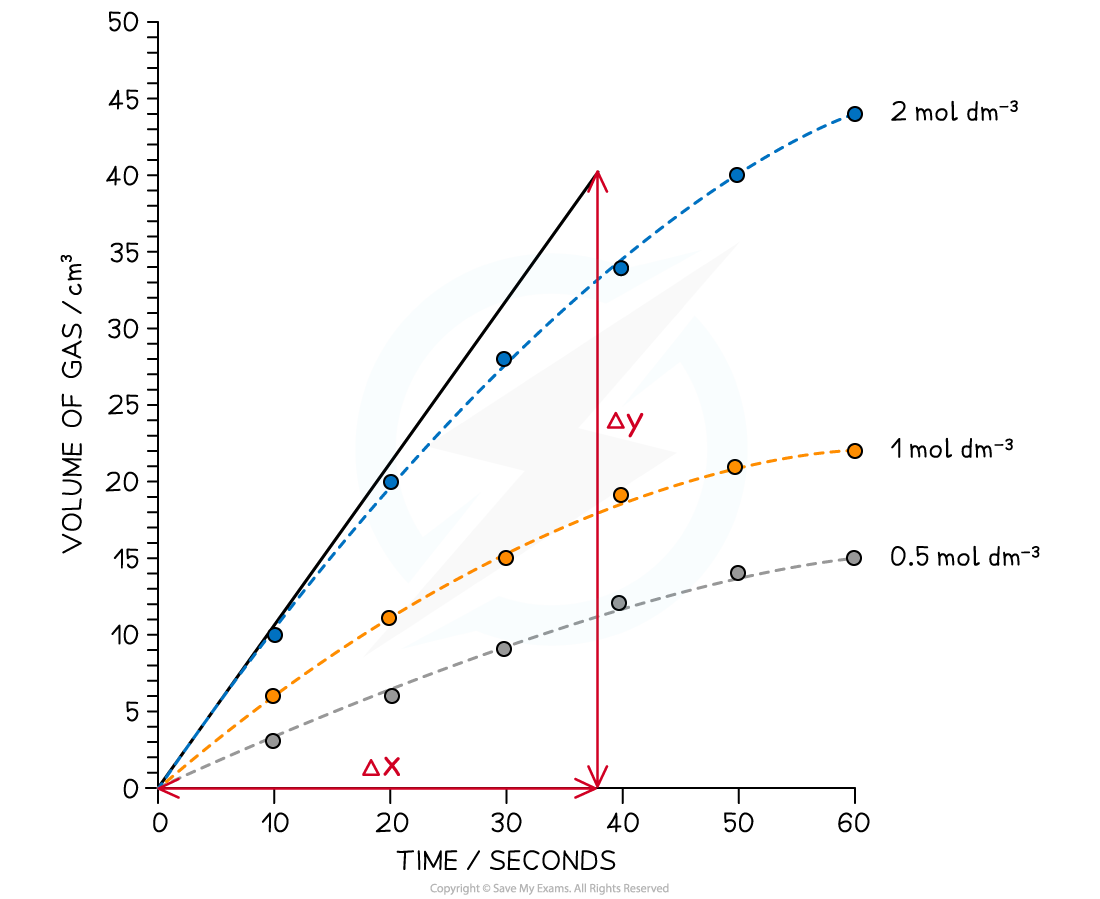
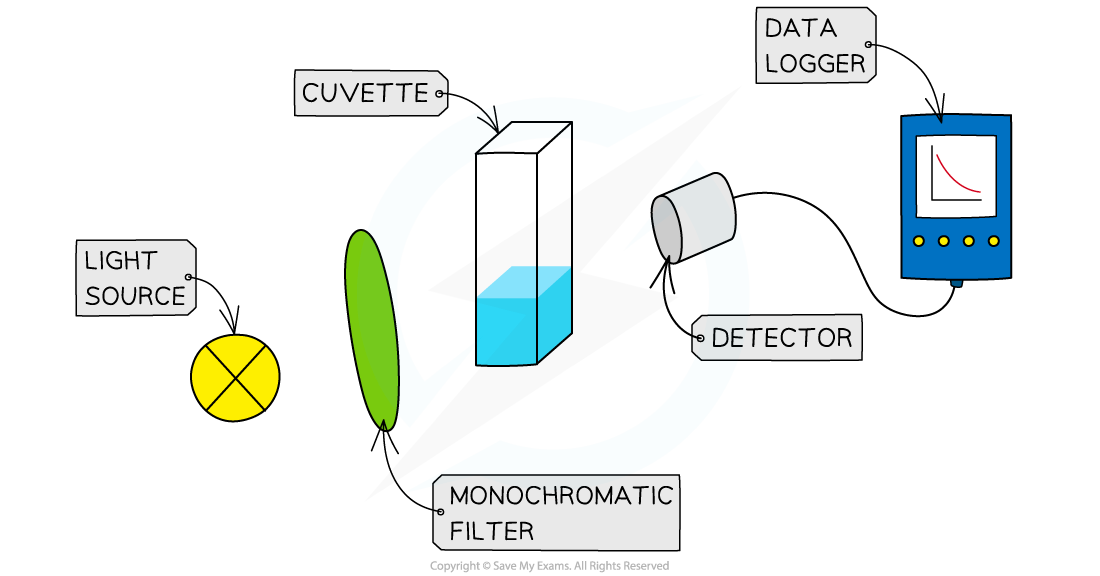
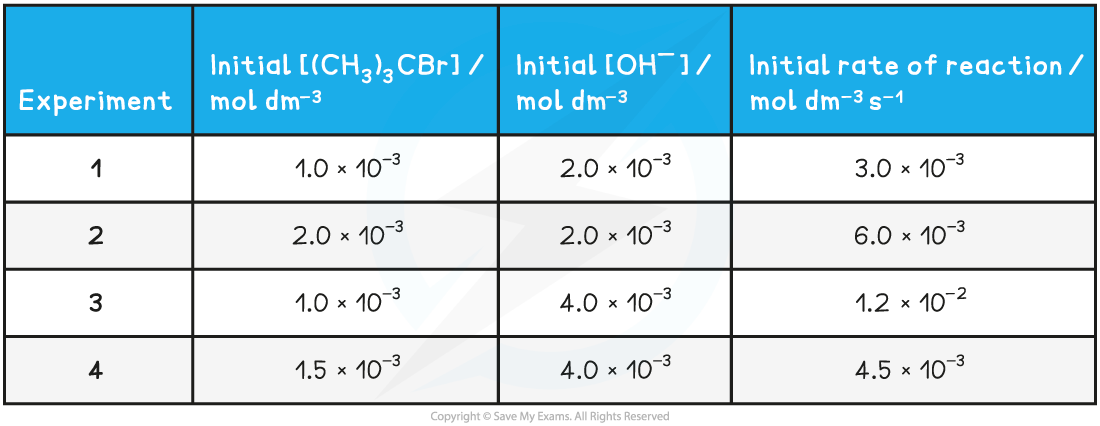
Reaction Order Graphs Reaction Order Using Concentration-Time Graphs In a zero-order reaction, the concentration of the reactant is inversely proportional to time This means that the reactan...