- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 400 888 0080
Edexcel A Level Chemistry:复习笔记1.5.2 Intermolecular Forces
Intermolecular Forces
Intramolecular forces
- Intramolecular forces are forces within a molecule and are usually covalent bonds
- Covalent bonds are formed when the outer electrons of two atoms are shared
- Single, double, triple and co-ordinate bonds are all types of intramolecular forces
Intermolecular forces
- Molecules also contain weaker intermolecular forces which are forces between the molecules
- There are three types of intermolecular forces:
- Induced dipole – dipole forces also called van der Waals or London dispersion forces
- Permanent dipole – dipole forces are the attractive forces between two neighbouring molecules with a permanent dipole
- Hydrogen Bonding are a special type of permanent dipole - permanent dipole forces
- Intramolecular forces are stronger than intermolecular forces
- For example, a hydrogen bond is about one tenth the strength of a covalent bond
- The strengths of the types of bond or force are as follows:
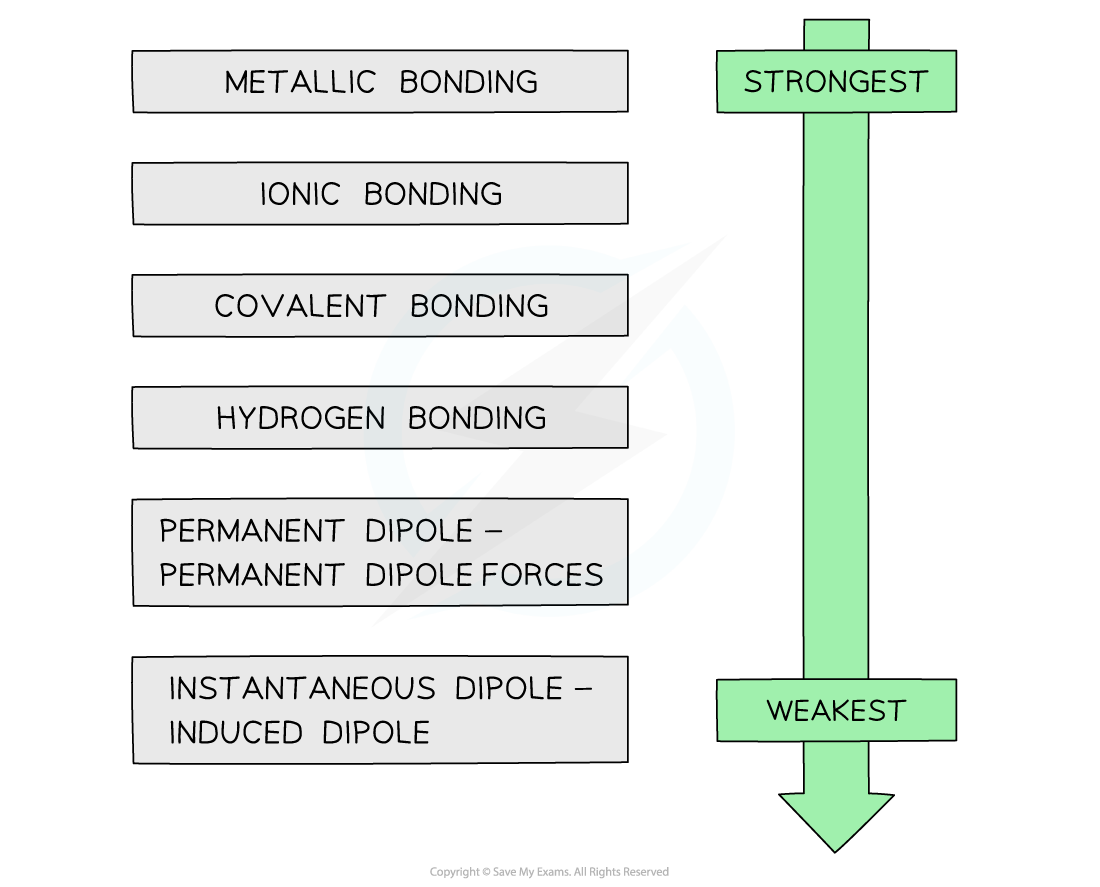
The varying strengths of different types of bonds
Induced dipole-dipole forces:
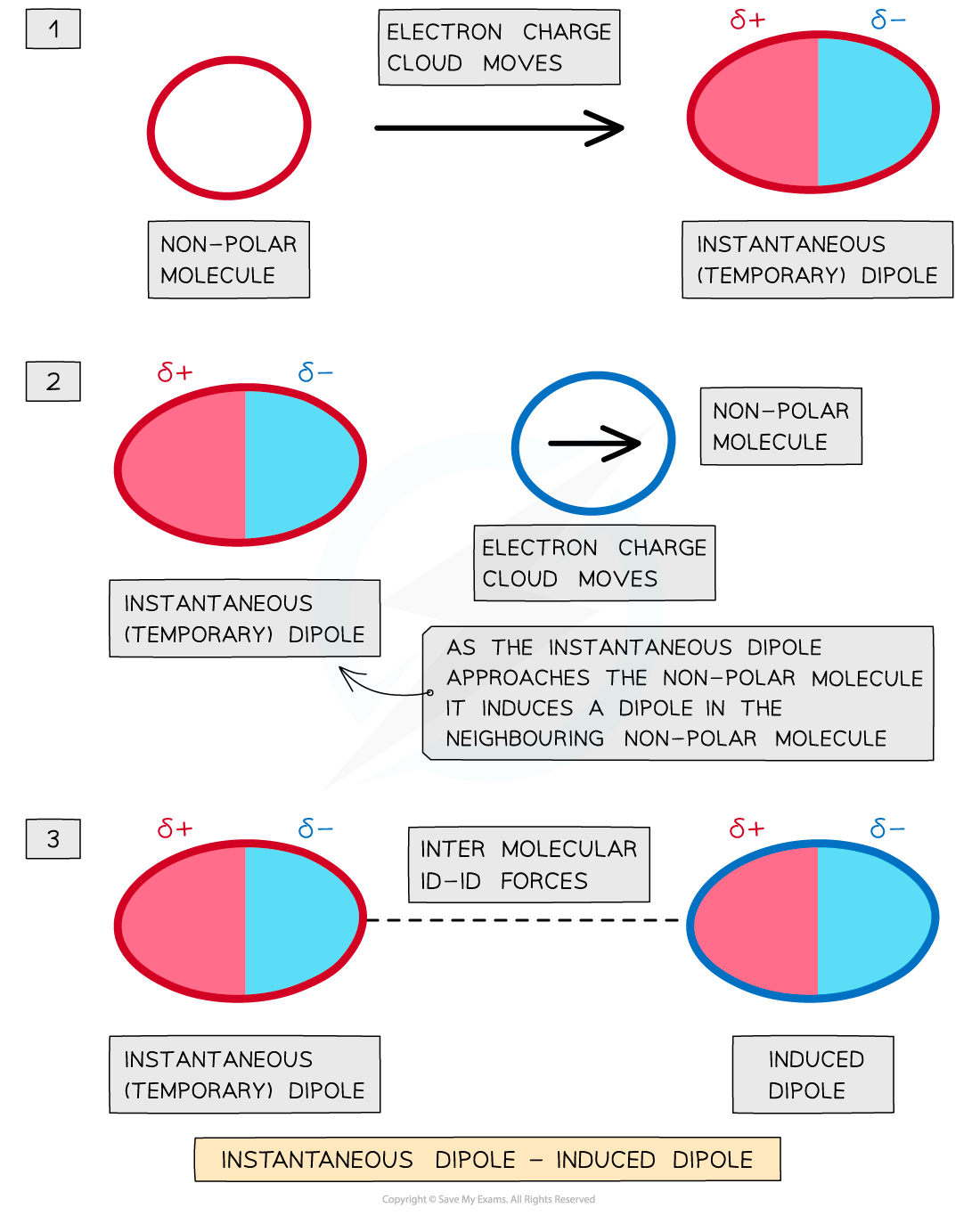
- Induced dipole - dipole forces exist between all atoms or molecules
- They are also known as London dispersion forces
- They are also known as London dispersion forces
- The electron charge cloud in non-polar molecules or atoms are constantly moving
- During this movement, the electron charge cloud can be more on one side of the atom or molecule than the other
- This causes a temporary dipole to arise
- This temporary dipole can induce a dipole on neighbouring molecules
- When this happens, the δ+ end of the dipole in one molecule and the δ- end of the dipole in a neighbouring molecule are attracted towards each other
- Because the electron clouds are moving constantly, the dipoles are only temporary
Relative strength
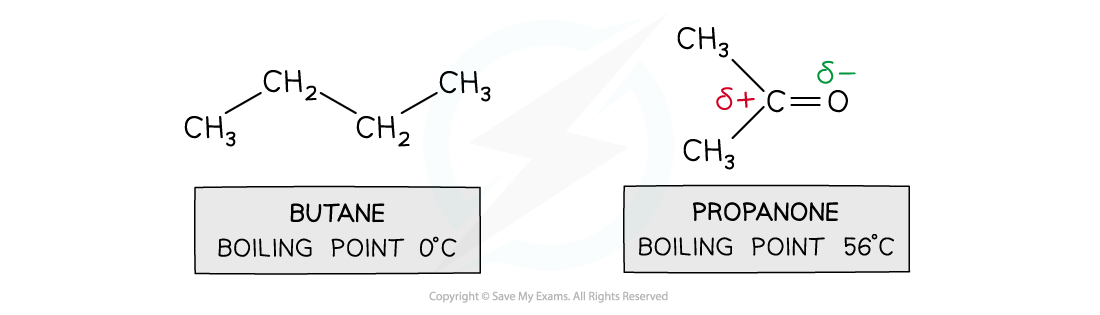
- For small molecules with the same number of electrons, permanent dipoles are stronger than induced dipoles
- Butane and propanone have the same number of electrons
- Butane is a nonpolar molecule and will have induced dipole forces
- Propanone is a polar molecule and will have permanent dipole forces
- Therefore, more energy is required to break the intermolecular forces between propanone molecules than between butane molecules
- So, propanone has a higher boiling point than butane
Pd-pd forces are stronger than id-id forces in smaller molecules with an equal number of electrons
Permanent dipole - dipole forces:
- Polar molecules have permanent dipoles
- The molecule will always have a negatively and positively charged end
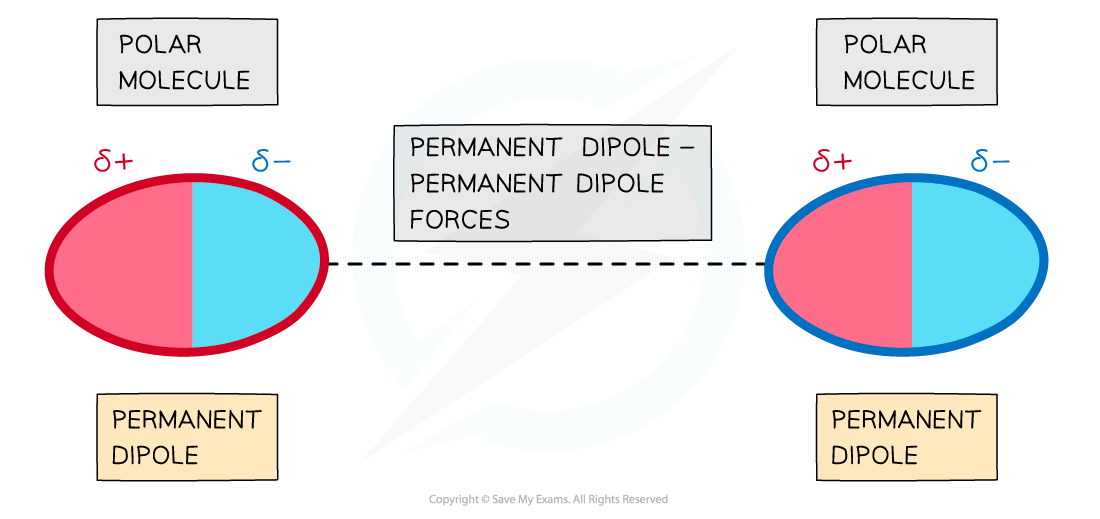
- Forces between two molecules that have permanent dipoles are called permanent dipole - dipole forces
- The δ+ end of the dipole in one molecule and the δ- end of the dipole in a neighbouring molecule are attracted towards each other
Hydrogen bonding
- Hydrogen bonding is the strongest form of intermolecular bonding
- Intermolecular bonds are bonds between molecules
- Hydrogen bonding is a type of permanent dipole – permanent dipole bonding
- For hydrogen bonding to take place the following is needed:
- A species which has an O, N or F (very electronegative) atom bonded to a hydrogen
- When hydrogen is covalently bonded to an O, N or F, the bond becomes highly polarised
- The H becomes so δ+ charged that it can form a bond with the lone pair of an O, N or F atom in another molecule
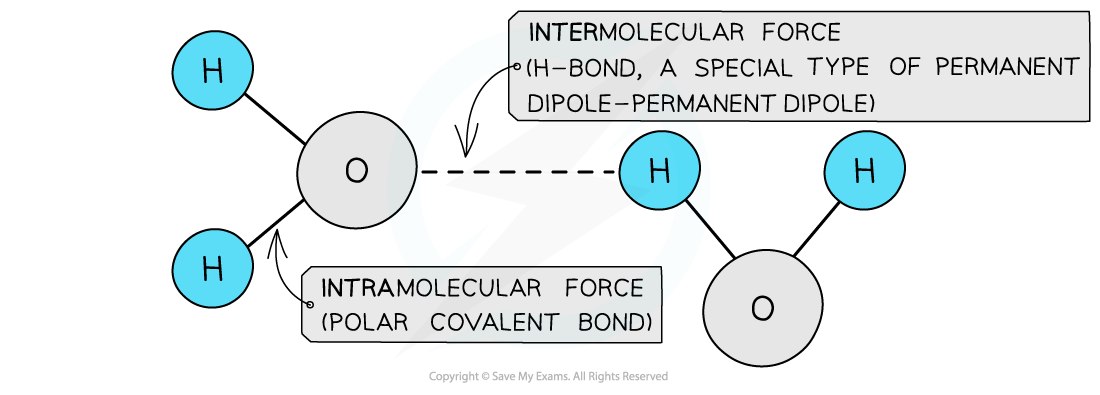
- For example, in water
- Water can form two hydrogen bonds, because the O has two lone pairs
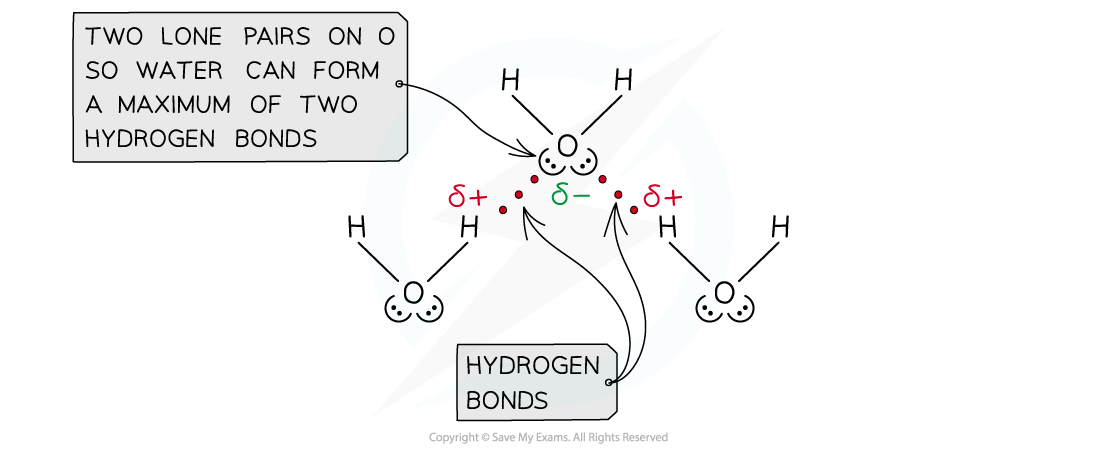
Hydrogen bonding in water
Exam Tip
Make sure to use a dashed, straight line when drawing your intermolecular forces! Hydrogen bonds should start at the lone pair and go right up to the delta positive atom - it must be really clear where your H bond starts and ends.
转载自savemyexams
早鸟钜惠!翰林2025暑期班课上线

最新发布
© 2025. All Rights Reserved. 沪ICP备2023009024号-1








