- 翰林提供学术活动、国际课程、科研项目一站式留学背景提升服务!
- 021-63526630
AQA A Level Biology复习笔记1.4.8 Limiting Factors Affecting Enzymes: pH
Rate: pH
- All enzymes have an optimum pH or a pH at which they operate best
- Enzymes are denatured at extremes of pH
- Hydrogen and ionic bonds hold the tertiary structure of the protein (ie. the enzyme) together
- Below and above the optimum pH of an enzyme, solutions with an excess of H+ ions (acidic solutions) and OH- ions (alkaline solutions) can cause these bonds to break
- This alters the shape of the active site, which means enzyme-substrate complexes form less easily
- Eventually, enzyme-substrate complexes can no longer form at all
- At this point, complete denaturation of the enzyme has occurred
- Where an enzyme functions can be an indicator of its optimal environment:
- Eg. pepsin is found in the stomach, an acidic environment at pH 2 (due to the presence of hydrochloric acid in the stomach’s gastric juice)
- Pepsin’s optimum pH, not surprisingly, is pH 2
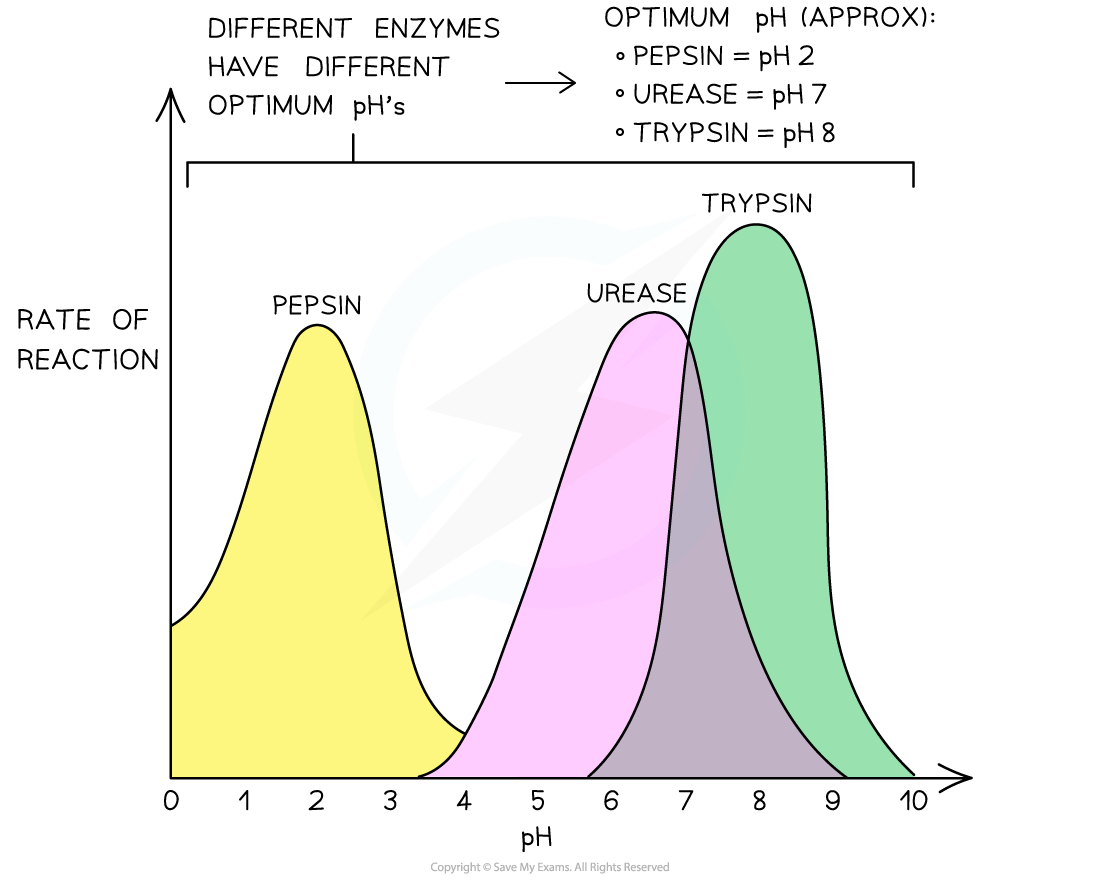
The effect of pH on the rate of an enzyme-catalysed reaction for three different enzymes (each with a different optimum pH)
- When investigating the effect of pH on the rate of an enzyme-catalysed reaction, you can use buffer solutions to measure the rate of reaction at different pH values:
- Buffer solutions each have a specific pH
- Buffer solutions maintain this specific pH, even if the reaction taking place would otherwise cause the pH of the reaction mixture to change
- A measured volume of the buffer solution is added to the reaction mixture
- This same volume (of each buffer solution being used) should be added for each pH value that is being investigated
转载自savemyexams
以上就是关于【AQA A Level Biology复习笔记1.4.8 Limiting Factors Affecting Enzymes: pH】的解答,如需了解学校/赛事/课程动态,可至翰林教育官网获取更多信息。
往期文章阅读推荐:
全网破防!ALevel CIE数学M1疑似错题?经济P2难度飙升?5月6日大考考情分析必看!
A-Level CIE就大规模泄题发布最严处罚!哪些考生必须重考?你的成绩怎么办?

最新发布
© 2026. All Rights Reserved. 沪ICP备2023009024号-1









